 The valence electrons are held closer towards the nucleus of the atom. This means that the nucleus attracts the electrons more strongly, pulling the atom's shell closer to the nucleus. 
The effect of increasing proton number is greater than that of the increasing electron number therefore, there is a greater nuclear attraction. However, at the same time, protons are being added to the nucleus, making it more positively charged. This is because, within a period or family of elements, all electrons are added to the same shell. Atomic radius patterns are observed throughout the periodic table.Ītomic size gradually decreases from left to right across a period of elements. The covalent radii of these molecules are often referred to as atomic radii. Nevertheless, it is possible for a vast majority of elements to form covalent molecules in which two like atoms are held together by a single covalent bond. Some are bound by covalent bonds in molecules, some are attracted to each other in ionic crystals, and others are held in metallic crystals. However, this idea is complicated by the fact that not all atoms are normally bound together in the same way. The atomic radius is one-half the distance between the nuclei of two atoms (just like a radius is half the diameter of a circle). This is caused by the increase in atomic radius. Electron affinity decreases from top to bottom within a group.This is caused by the decrease in atomic radius. Electron affinity increases from left to right within a period.This causes the electron to move closer to the nucleus, thus increasing the electron affinity from left to right across a period. Moving from left to right across a period, atoms become smaller as the forces of attraction become stronger. With a larger distance between the negatively-charged electron and the positively-charged nucleus, the force of attraction is relatively weaker. This means that an added electron is further away from the atom's nucleus compared with its position in the smaller atom. It is important to recognize which numbering system is being used and to be able to find the number of valence electrons in the main block elements regardless of which numbering system is being used.\( \newcommand\): Periodic Table showing Electron Affinity TrendĮlectron affinity generally decreases down a group of elements because each atom is larger than the atom above it (this is the atomic radius trend, discussed below). You will come across periodic tables with both numbering systems. The outer electron shell is not filled for these elements, giving them a valence. 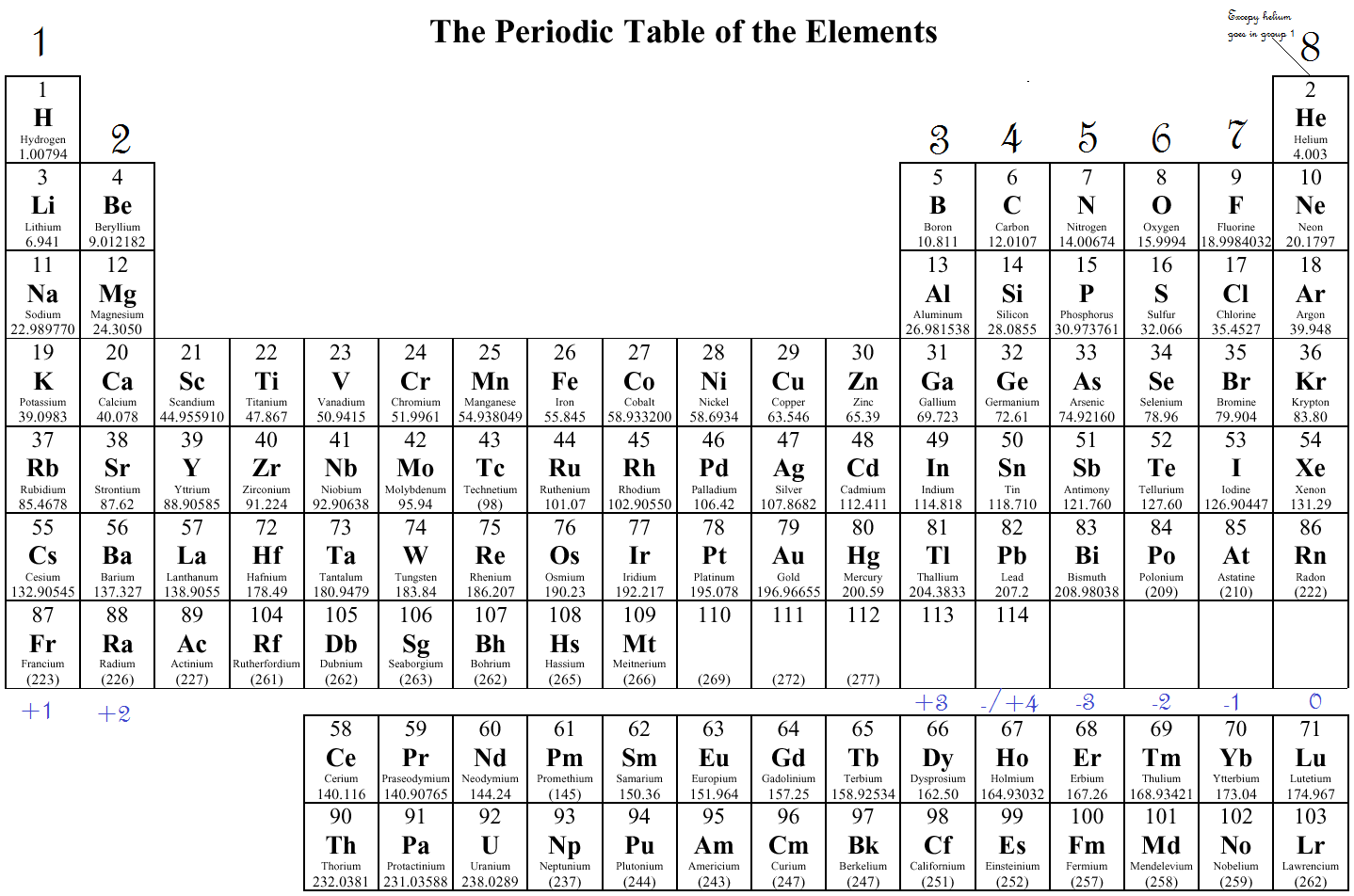
The representative elements are groups 1 and 2 and group 13-17 on the periodic table. Another name for the representative elements is the main group elements. In this numbering system, group 1A is group 1 group 2A is group 2 the halogens (7A) are group 17 and the noble gases (8A) are group 18. In chemistry, the representative elements are the elements with atoms filling s and p electron orbitals. The elements in this group are also gases at room temperature.Īn alternate numbering system numbers all of the \(s\), \(p\), and \(d\) block elements from 1-18. We will learn the reason for this later, when we discuss how compounds form. These elements also have similar properties to each other, the most significant property being that they are extremely unreactive, rarely forming compounds. This group contains very reactive nonmetal elements. Group 7A (or 17) elements are also called halogens. It is important to recognize a couple of other important groups on the periodic table by their group name. Remember, Mendeleev arranged the table so that elements with the most similar properties were in the same group on the periodic table. The same pattern is true of other groups on the periodic table. Once again, because of their similarities in electron configurations, these elements have similar properties to each other. Group 2A is also called the alkaline earth metals. Note the definition of an orbital is the defined regions of space where electrons can be found 95 percent of the time. each of these orbitals is shaped differently, and depict the way an electron might float around the nucleus. Orbitals come in different shapes: s, p, d, f. Although most metals tend to be very hard, these metals are actually soft and can be easily cut. Electrons can be anywhere inside the electron cloud at any moment in time. Group 1A is also known as the alkali metals. Because of their similarities in their chemical properties, Mendeleev put these elements into the same group. The elements in 1A are all very reactive and form compounds in the same ratios with similar properties with other elements. This is what causes these elements to react in the same ways as the other members of the family. All of the 1A elements have one valence electron. A group is a vertical column of the periodic table. 
Remember that Mendeleev arranged the periodic table so that elements with the most similar properties were placed in the same group. 
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
